Abstract
In order to develop rearing techniques for Marphysa sanguinea Montagu (1813), the influence of salinity on survival and growth rates of juveniles has been investigated under controlled laboratory conditions. For this purpose, 720 worms were produced in the laboratory. After rearing up to 6–9 setigerous segments, the worms were subjected to an experimental set-up for 2 months, in which they were exposed to salinities ranging from 15 to 40. For each salinity set-up, two sediment groups were used: an unmodified natural sediment and the same supplemented with Ulva lattuca, an assumed important natural dietary item. The results obtained showed the following: (1) Salinity changes produce an immediate and significant effect on growth; (2) no significant differences were noted between the two groups subjected to different diets, but a significant interaction was noted between each of the two factors. Results of this study can have significant implications for M. sanguinea aquaculture, concerning farm site selection and salinity maintenance in order to maximize commercial productivity.
Similar content being viewed by others
Introduction
Environmental factors strongly affect all phases of the life cycle of many marine invertebrates, and in particular of species inhabiting coastal and estuarine regions.
Temperature and salinity are primary abiotic variables affecting survival, activity and distribution of marine organisms (Kinne 1964), and environmental tolerance to salinity may not be the same for gametes as for adults (Fong 1998). Most euryhaline animals are not immune to the effects of salinity changes. Documented sublethal effects of altered salinity include reduced rates of growth and development (Qiu and Qian 1997 ; Lambert et al. 1994; Rosas et al. 1999; Specker et al. 1999) and reduced fecundity (Morritt and Stevenson 1993; Pechenik et al. 2007).
The effects of diet, sediment organic content, pollutants and prolonged larval life on growth and life history characteristics have been well documented for capitellid species (e.g. Pechenik and Cerulli 1991; Qian and Chia 1992a,b; Bridges 1996; Levin et al. 1996; Cohen and Pechenik 1999), but the effects of salinity on survival and somatic development of the Eunicidae have not been reported previously.
The iteroparous and long-lived polychaete M. sanguinea has been recorded from many localities around the world, often without an exhaustive description or deposition of voucher material. Recently, there has been some debate on whether M. sanguinea from different geographical regions may in fact encompass a series of cryptic species, morphologically difficult to distinguish (Hutchings and Karageorgopoulos 2003; Lewis and karageorgopoulos 2008).
In Portugal, the species is present along the Sado estuary, located in the south of the Setúbal peninsula, 25 km south-east of Lisbon (Fig. 1), particularly within former oyster production areas. In this estuary, M. sanguinea lives in deep borrows, 25–40 cm into the intertidal mudflats. It is a polychaete of considerable size, attaining a maximum length of about 40–50 cm and is a gonochoric species with an annual iteroparous reproductive strategy. According to Castro (1993), it has a contagious distribution, spreading in aggregates of different sizes. Concerning the dietary habits, the same author, through an analysis of the digestive contents, observed that this worm ingests preferentially benthic macroalgae (Ulva, Enteromorpha, Fucus vesiculosos) as well as fine sediment with benthic microalgae normally abundant in the intertidal mudflats of the estuaries.
Potential uses
The commercial interest in polychaetes mainly arises from their value as bait for recreational fishing and as food source for penaeid crustaceans and finfish in aquaculture (Dinis 1986; Olive 1994). This has lead to the development of small but economically viable aquaculture facilities providing a supply of different species (Gambi et al. 1994; Olive et al. 2000) for a variety of uses. Simultaneously, traditional bait digging exploitation continues to occur, presenting an ill-defined boundary between anglers collecting strictly for their own use and those collecting for direct profit. The activity therefore represents a form of parallel economy, directly conflicting with a sustainable and controlled use (Olive 1993).
M. sanguinea is commonly known in Portugal by the names of “ganso” and “minhocão”. It is one of the most appreciated bait species among those collected and commercialized in Portugal due to the high resistance it presents on the hook and to its reputed ability to attract valuable fish species such as the seabreams Sparus aurata and Diplodus sargus. Its harvest activity is one of the most important socio-economic resources for local fishermen. The price per litre of “minhocão” in 2007 ranged between 150 and 200 Euros, and in 2008 between 250 and 300 Euros, and it is estimated that between 2007 and 2009 the annual sales volume of the “minhocão” in the Sado estuary has fluctuated between 600 and 750 thousand Euros (Directorate General for Fisheries and Aquaculture -DGPA-, personal communication). More exact quantification is difficult because a substantial component is sold through a parallel economy in which sales are not declared.
The long-term exploitation of this species requires a good knowledge of biological and ecological characteristics of the natural populations that may allow optimized harvest and minimized collection impact. The main aim and the scientific challenge of the research on the biology of this bait polychaete species should be to develop rearing techniques for intensive production that may increase resource supply and potentially reduce harvest and its impact on natural populations. However, the same acquired knowledge can also serve to provide the tools for a sustainable utilization of the resource form the wild.
With this purpose in mind, a small-scale experiment was conducted in order to elucidate an important aspect of the role of salinity on the life cycle of M. sanguinea and more specifically the salinity level most suitable to its somatic growth. Further development of polychaete culture is considered of economic importance beyond that associated with the bait supply industry which acted as the initial catalyst for this research.
Materials and methods
In March 2006, 720 juveniles were produced in the laboratory from a population collected from the Sado estuary. At six to nine setigerous segments, their length was determined from the measurement of the width of the buccal segment (W.S.B.) (Fig. 2) according to Castro (1993). They were thence used for 2 months to study the development and growth rate in relation to salinity under two diet regimes.
Dorsal view of the anterior region of M. sanguinea, adapted from Castro (1993). W.S.B—Width of buccal segment
The experimental set-up consisted in placing the animals in 36 containers (1 L) and maintaining them at six different salinities (15, 20, 25, 30, 35 and 40) in a laboratory (22 ± 0.5°C) with constant 12/12 light/dark photoperiod. All the containers were filled with a fine layer of sediment (1 cm) collected from the natural environment of the animals along the Marateca channel. The sediment was filtered through a 0.5-mm sieve in order to remove macroalgae and any associated macrofauna, and it was then placed in an oven at 90°C for 24 h and first divided into 6 groups according to the salinity range. For each salinity treatment, one half of the containers received an Ulva lattuca supplement. A population of 20 worms was placed randomly in each of the recipients, resulting in three replicates for each salinity and sediment type. The used water was renewed daily. Before adding to the recipients, new water was filtered through a biological filter and a cartridges series (<10, <5 and <1 μm) and maintained under constant oxygenation. During the first 48 h, the polychaetes were allowed to settle (construct and establish burrows). U. lattuca were provided daily, fresh and “ad libitum” after this period. For the biometric analysis, three recipients from each salinity were randomly selected weekly. While taking the measurements, worms were extracted and the recipients were placed back in the circuits in order to keep the flows constant. Length was always determined from the number of setigers formed and from the W.S.B. The linear measurements of W.S.B obtained were taken under a stereomicroscope equipped with a micrometric ocular at 2× magnification. The dimensions obtained were processed by the image-processing software system Qwin Lite 2.4 (Leica Mycrosystems Imaging Aquisition). Statistical analysis was performed using SPSS (statistical package for the social sciences) software (version 14; SPSS Inc, Chicago, Il). The relationships between salinities and subtracts and the effects of salinity and subtract on growth were analysed using a non-parametric two-way analysis of variance (ANOVA), followed by a multiple comparison of means (LSD), when the treatment means showed significant differences. A significance level of α = 0.05 was chosen.
Results
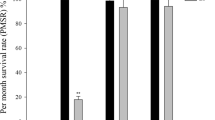
Although juvenile survival was generally high in all the salinities tested, the worms reared at 15, 20 and 40 displayed more elevated mortalities than the remaining ones in which the mortality remained more or less constant throughout the experiment. Under the experimental conditions, mortality was only relatively high under the lowest and highest salinities in the range (15, 40) in the first weeks of rearing and then tended to level off. No differences exclusively attributable to U. lattuca were observed (Fig. 3).
Survival, size patterns, and setigerous segment development of Marphysa sanguinea juveniles at each of a series of salinity and two sediment treatments conducted over 8 weeks. Numbers on top of each panel correspond to salinity values. Length was determined by the difference between the total length (TL) obtained weekly (TLW) and the initial total length (TL0): Length = TLW−TL0. Length was estimated from the width of the buccal segment (W.S.B.) from the equation TL = 3.9303WSB 1.3307 according to Castro (1993)
At the start of the experiment, the animals measured 0.19 ± 0.2 cm.
Throughout the experiment, no significant differences in growth were obtained among replicates. However, significant differences (P < 0.01) were observed in both the development and the growth rates of M. sanguinea juveniles influenced by different salinities, whereas no effect was noted between the two different sediment types, but a significant interaction was found between them (Table 1). The comparison of means with the LSD test indicates that salinity effects can be individualized in three groups with different influences (P < 0.05): in the first one, constituted by the worms reared at 15, 20 and 40, growth was usually reduced (P < 0.05 and P < 0.01) when compared to 25, 30 and 35, respectively. In fact, after 4 weeks (see Fig. 3), juveniles maintained in salinity 15 practically did not grow, reaching a length of 0.4 ± 0.2 cm in both sediment set-ups. At 20 and 40, growth was still low (0.7 ± 0.4 cm; 0.9 ± 0.4 cm and 0.7 ± 0.2 cm; 0.4 ± 0.2 cm in length, respectively), which contrasts with the results obtained at 30 (1.4 ± 0.6 cm; 1.7 ± 0.4 cm) and 35 (1.3 ± 0.6 cm; 1.6 ± 0.6 cm). In fact, at 30 and 35, the worms grew more in 4 weeks than during the whole experiment at the other salinity treatments. On the other hand, the results obtained at 25 (1 ± 0.5 cm; 1.2 ± 0.5 cm) suggest that this salinity represents an intermediate level between the upper and lower salinities.
The results previously discussed seem supported when the number of setigers developed is considered. (see Fig. 3). However, here, well-defined extremes and a clear preference for U. lattuca exist mainly at salinities 25, 30 and 35 where growth rates are high. In fact, it is clear that at salinities 15 (18 ± 3; 20 ± 2 setigers) and 35 (33 ± 4; 40 ± 4 setigers), respectively, lie the worst and the best suitable conditions observed in the experiments when this developmental feature is considered. In the group reared at 20 and 40, the worms developed 22 ± 4; 24 ± 4 and 23 ± 3; 25 ± 3, respectively, and at 25 and 30 after 8 weeks, the worms reached 30 ± 3; 34 ± 4 and 34 ± 4; 36 ± 3, respectively. A significant linear correlation (P < 0.001) between total length and number of setigers formed was obtained. Morphological development appears consistent with growth rates. In worms exposed to 15, 20 and 40, the jaws are well developed, and 4 eyes, capillary setae, 4 antennae and simple gills appeared at the end of the experiment. However, worms exposed to salinities 25, 30, and 35 at 4 weeks showed well-developed jaws, 2 pairs of eyes and 5 antennae, and at the end of the experiment, a red pigmentation and well-developed gills were clearly visible from the 10th segment extending out to the 30th.
Discussion
Almost all polychaetes are known to be euryhaline, although salinity tolerance has not previously been well defined for M. sanguinea.
This study showed that juveniles of this species are very resistant to salinity fluctuations. In fact, the largest mortality occurred in the first week, which suggests it may be related with adaptation to laboratory conditions and not to the experimental treatments. On the other hand, juveniles exhibit a very territorial and aggressive behaviour with high rates of cannibalism if the environmental conditions are disadvantageous (pers. obs.), so that the mortality under experimental conditions may also derive from cannibalism and not directly from the conditions. It also appears that the tolerance of the juveniles of M. sanguinea to different salinities changes with age, so that differential survival along the experiment may derive to an extent from the changing tolerance. Therefore, mortality patterns remain largely unexplained and require further research.
Growth rates, on the other hand, were significantly (P < 0.05) influenced by salinity, and although there was not a clear preference for U. lattuca, growth rates under a diet supplemented by the alga were higher at all salinities, indicating a clear benefit accrued from the diet. Prevedelli (1994) in Marphysa juveniles and Pechenik et al. (2000) in Capitella juveniles obtained a significantly higher growth at salinity of 30. The influence of salinity on growth has also been demonstrated in other species. For example, at low salinity Nereis diversicolor has a greater growth rate than Nereis succinea (Neuhoff 1979). It is also known that under laboratory conditions, a few aquatic invertebrates can attain a maximum growth rates at salinity levels which are lower or higher than those of natural waters (Kinne 1971).
The significant correlation (P < 0.001; R 2 = 0.86) between total length and the number of setigerous segments obtained in this study is in agreement with previous findings reporting setigerous development to be a good parameter for estimating total length of juveniles. However, this parameter cannot always be used in older stages or adults due to the elevated autotomy that occurs in this species (Fauvel 1923; Castro 1993).
M. sanguinea, similarly to the majority of the known polychaetes, colonizes brackish waters. In the Sado estuary, colonies are found in biotypes of high and low natural salinities; more precisely, they are found in the Marateca (about 10% freshwater inflow—>36 at times) and Alcácer (80% freshwater inflow—<15 at times) channels (see Fig. 1). It has been shown that there are different degrees of adaptation to salinity variations. According to Abbiati (1996), N. diversicolor is the Mediterranean species that shows the highest genetic adaptation to brackish environments, but adaptation to salinity fluctuation has been found to be maximum in Nereis limnicola due to a strategic reproductive feature that protects larvae from the lowest salinities (Smith 1957, 1964). Krishnamoorthi (1951) demonstrated experimentally that the gelatinous masses observed in Marphysa gravelyi provide the eggs and embryos with an osmotic protection under conditions of salinity fluctuation. Garcês (2002) also observed in the laboratory in spawns that occurred in galleries inside oyster shells that the eggs of M. sanguinea were encapsulated in a gelatinous mass and protected by a jelly mucous forming “cocoon” where the larval development occurred. Aiyar (1931) observed in India a maximum reproductive activity in M. gravelyi after the monsoon season. Thus, conditions of low and variable salinity may serve to stimulate spawning in these species, possibly in anticipation of favourable trophic conditions, and the gelatinous masses may then provide the necessary environment in which embryonic development can proceed amidst a generally unfavourable environment.
We suggest that this species colonizes brackish waters and has a high capacity to tolerate variations in environmental parameters, in particular salinity, but that the most favourable developmental conditions lie within relatively limited parameters as shown.
Conclusion
The present experiment, which was conducted under laboratory conditions, utilizing specimens hatched in the laboratory from eggs layed by mature worms collected in Sado estuary demonstrates that in M. sanguinea juveniles, salinity can have an immediate and significant effect on growth. A salinity range outside 25–35 will significantly reduce growth rates. Salinities close to 15 and 40 seem to be the lower and upper physiological limit for this species. Thus, a salinity range between 30 and 35 is recommended for the juvenile culture of this polychaete.
The results obtained in this study have permitted a better understanding of larval development and population dispersion patterns of M. sanguinea juveniles in the Sado estuary and possibly elsewhere, but also bear significant implications for the aquaculture of the species, concerning farm site selection and salinity maintenance in order to maximize commercial productivity.
References
Abbiati M (1996) Genetic adaptation to brackish environment: an example from the family Nereididae (Annelida: Polychaeta). In: Abstracts of the 31rd EMBS, St. Petersburg, Sep 1996, p 12
Aiyar RG (1931) An account of the development and breeding habits of a brackish-water polychaete worm of the Genus Marphysa. J Linn Soc (Zool) 37:387–403
Bridges TS (1996) Effects of organic additions to sediment, and maternal age and size, on patterns of offspring investment and performance in two opportunistic deposit-feeding polychaetes. Mar Biol 125:345–357
Castro JJ (1993) Impacte da exploração comercial e estrutura, dinâmica e produção da população de Marphysa sanguinea (Annelida: Polychaeta) no estuário do Sado. Dissertation, University of Évora
Cohen RA, Pechenik JA (1999) Relationship between sediment organic content, metamorphosis, and postlarval performance in the deposit-feeding polychaete Capitella sp I. J Exp Mar Biol Ecol 240:1–18
Dinis MT (1986) Quatre Soleidae de l'estuaire du Tage: reproduction et croissance essai d'elevage de Solea senegalensis Kaup. PhD Thesis, University of Bretagne Occidentale, Brest
Fauvel P (1923) Polychètes errantes. Faune de France 5:488
Fong PP (1998) Zebra Mussel spawning is induced in low concentrations of putative serotonin reuptake inhibitors. Biol Bull 194:143–149
Gambi MC, Castelli A, Giangrande A, Lanera P, Prevedelli D, Zunarelli Vandini R (1994) Polychaetes of commercial and applied interest in Italy: an overview. In: Dauvin JC, Laubier L, Reish, DJ (eds) Actes de la 4ème Conférence Internationale des Polychètes. Mem Mus Nat Hist Nat (Fr) 162: 593–603
Garcês JP (2002) Contribution for the study of Marphysa sanguinea Montagu (Polychaeta:Eunicidae). In: Abstracts of the 12rd Iberian Symposium on Studies of Marine Benthos, La Línea, Gibraltar, 22–25 Oct, p 78
Hutchings PA, Karageorgopoulos P (2003) Designation of a neotype of Marphysa sanguinea (Montagu, 1813) and a description of a new species of Marphysa from eastern Australia. Hydrobiologia 496:87–94
Kinne O (1964) The effects of temperature and salinity on marine and brackish water animals: II. Salinity and temperature salinity combinations. Oceanogr Mar Biol Annu Rev 2:281–339
Kinne O (1971). In: Kinne O (ed) Marine ecology: a comprehensive, integrated treatise on life in oceans and coastal waters, vol 1, part 2. Wiley-Interscience, Wiley and Sons, New York, pp 821–954
Krishnamoorthi B (1951) Studies on osmotic proprieties of the eggs and larvae of a brackish-water polychaete, Marphysa gravelyi Southern. Proc Indian Acad Sci 34:199–209
Lambert Y, Dutil JD, Munro J (1994) Effects of intermediate and low salinity conditions on growth rate and food conversion of Atlantic cod (Gadus morhua). Can J Fish Aquat Sci 51:1569–1576
Levin L, Caswell HL, Bridges T, DiBacco C, Cabrera D, Plaia G (1996) Demographic responses of estuarine polychaetes to pollutants: life table response experiments. Ecol Appl 6:1295–1313
Lewis C, Karageorgopoulos P (2008) A new species of Marphysa (Eunicidae) from the western Cape of South Africa. J Mar Biol Assoc UK 88(2):277–287
Morritt D, Stevenson TDI (1993) Factors influencing breeding initiation in the beachflea Orchestia gammarellus (Pallas) (Crustacea: Amphipoda). J Exp Mar Biol Ecol 165:191–208
Neuhoff HG (1979) Influence of temperature and salinity on food conversion and growth of different Nereis species (Polychaeta: Annelida). Mar Ecol Prog Ser 1:255–262
Olive PJW (1993) Management of the exploitation of the lugworm Arenicola marina and the ragworm Nereis virens (Polychaeta) in conservation areas: the importance of population structure and recruitment processes. Aquat Conserv 3:1–24
Olive PJW (1994) Polychaeta as a world resource: a review of patterns of exploitation as sea hangling baits and the potential for aquaculture based production. Mem Mus Natn Hist Nat 162:603–610
Olive PJW, Islam M, Cowin PBD (2000) Cultured polychaete: a dietary resource to increase penaeid hatchery performance. Eur Aquac Soc Spec publ 28:523
Pechenik JA, Cerulli TR (1991) Influence of delayed metamorphosis on survival, growth, and reproduction of the marine polychaete Capitella sp I. J Exp Mar Biol Ecol 151:17–27
Pechenik JA, Berard R, Kerr L (2000) Effects of reduced salinity on survival, growth, reproductive success, and energetics of thr euryhaline polychaete Capitella sp. I. J Exp Mar Biol Ecol 254:19–35
Pechenik JA, Pearse SJ, Qian PY (2007) Effects of salinity on spawning and early development of the tube-building polychaete Hydroides elegans in Hong Kong: not just the sperm’s fault? Biol Bull 212:151–160
Prevedelli D (1994) Influence of temperature and diet on the larval development and growth of juveniles Marphysa sanguinea (Montagu) (Polychaeta, Eunicidae). In: Dauvin JC, Laubier L, Reish DJ (eds) Actes de la 4ème Conférence internationale des Polychètes. Mémoire du Muséum National d’Histoire Naturelle 162: 521–562
Qian PY, Chia FS (1992a) Effect of aging on reproduction in a marine polychaete Capitella sp. J Exp Mar Biol Ecol 156:23–38
Qian PY, Chia FS (1992b) Effects of diet type on the demographics of Capitella sp. (Annelida: Polychaeta): lecithotrophic development vs. planktotrophic development. J Exp Mar Biol Ecol 157:159–179
Qiu JW, Qian PY (1997) Combined effects of salinity, temperature and food on early development of the polychaete Hydroides elegans. Mar Ecol Prog Ser 152:79–88
Rosas C, Ocampo L, Gaxiola G, Sánchez A, Soto LA (1999) Effect of salinity on survival, growth and oxigen consumption of postlarvae (PL10-PL21) of Litopenaeus setiferus. J Crus Biol 19:244–251
Smith RI (1957) A note on the tolerance of low salinities by Nereid polychaetes and its relation to temperature and reproductive habitat. Ann Biol 33:93–107
Smith RI (1964) On the early development of Nereis diversicolor in different salinities. J Morphol 114:437–464
Specker JL, Schreiber AM, McArdle ME, Pohoilek A, Henderson J, Bengtson DA (1999) Metamorphosis in summer flounder: effects of acclimation to low and high salinities. Aquaculture 176:145–154
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Garcês, J.P., Pereira, J. Effect of salinity on survival and growth of Marphysa sanguinea Montagu (1813) juveniles. Aquacult Int 19, 523–530 (2011). https://doi.org/10.1007/s10499-010-9368-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-010-9368-x